|
Although the use of ASPCs is best suited to the isolation of high affinity mAbs, since they go through the processes of somatic hypermutation and affinity maturation, the application of this method for species other than humans and mice is limited because the current plasma/plasmablast cell (PC) isolation protocols rely on a small number of identified PC-specific markers combined with the absence of one or more B cell differentiation antigens. Recently, the direct molecular cloning of cognate pairs of immunoglobulin gamma heavy chain (IgH) variable (V H), light chain kappa variable (V Lκ) and light chain lambda variable (V Lλ) genes from single antigen-specific plasma/plasmablast cells (ASPCs) using the polymerase chain reaction (PCR) has attracted attention as an alternative method for generating mAbs from immunized animals. While a variety of animals have been used to produce polyclonal antibodies against human proteins, mAbs from animals other than rodents have not been routinely produced due to the difficulties in establishing immortalized antibody-producing cell lines by hybridoma, viral transformation or reprogramming. To avoid the problem of human proteins being recognized as self-antigens in mice, the use of an evolutionarily distant animal from humans is essential to obtain better immunization against therapeutic target molecules. However, immune responses against highly conserved human proteins are often weak in mice, resulting in the production of low affinity and/or non-specific mAbs. The mouse hybridoma method has been used previously for the production of candidate monoclonal antibodies (mAbs) for therapeutic use. Our technology eliminates the need for both cell propagation and screening processes, offering a significant advantage over hybridoma and display strategies. Amplification of cognate pairs of immunoglobulin heavy and light chain genes followed by DNA transfection into 293FT cells resulted in the highly efficient production of antigen-specific monoclonal antibodies from a variety of immunized animals. By using a fluorescently labeled antigen as a tag for a complementary cell surface immunoglobulin, antigen-specific plasma/plasmablast cells were sorted from the rest of the cell population by fluorescence-activated cell sorting. 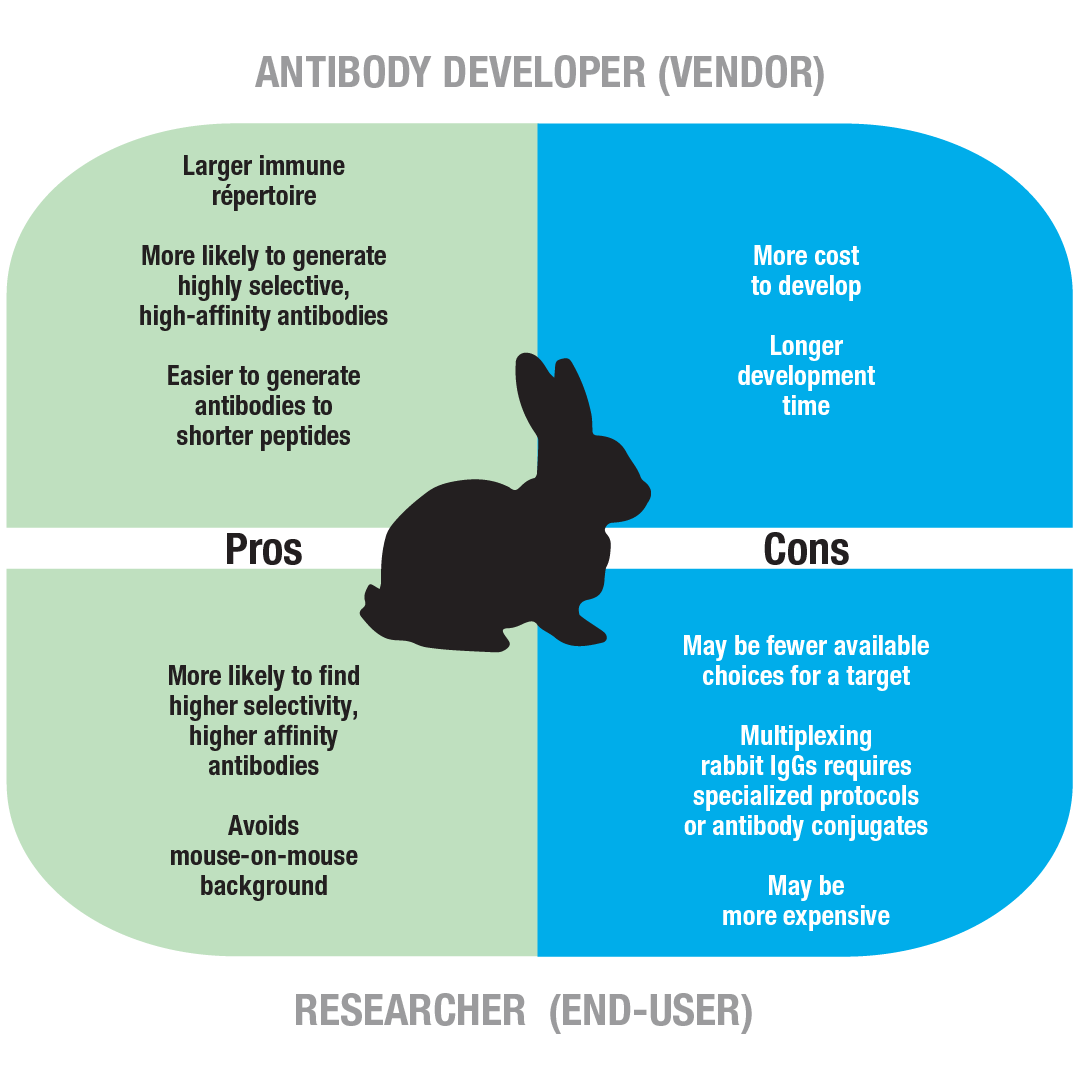 
By staining lymph node cells with an antibody against immunoglobulin and a fluorescent dye specific for the endoplasmic reticulum, plasma/plasmablast cells were identified without using a series of antibodies against lineage markers. We propose a simple and rapid strategy to produce monoclonal antibodies from a variety of animals. Although a variety of animals have been used to produce polyclonal antibodies against antigens, the production of antigen-specific monoclonal antibodies from animals remains challenging.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
